Explore the Map by Accessing the CCTAM
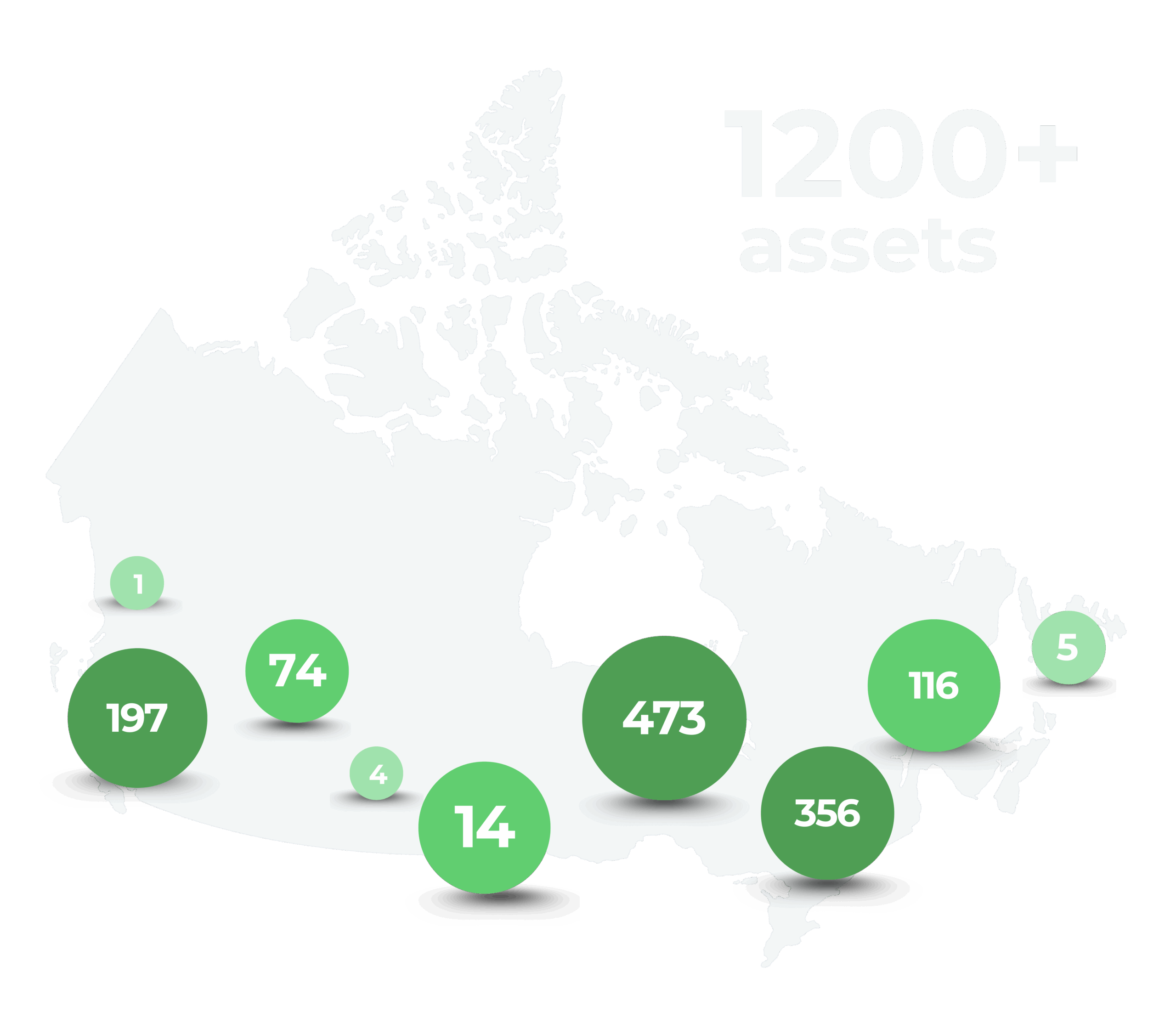

Join the Canadian Clinical Trials Asset Map (CCTAM), a national platform that connects sponsors, sites, and investigators to drive Canadian clinical research forward!

Why Join CCTAM?
Every day, sponsors search for Canadian sites and investigators to run trials. But too often, your capabilities remain hidden. CCTAM helps change that.
Create a free profile to:
- Boost your visibility with global sponsors and CROs
- Connect with peers across the country for collaboration
- Open doors to new trials and career-building opportunities
- Support Canada’s reputation as a global clinical research leader
What Is CCTAM?
The Canadian Clinical Trials Asset Map is a free, national, online database of clinical research assets, including trial sites, investigators, institutions, REBs, and networks across Canada.
Whether you’re running a large academic study or a community-based trial site, CCTAM is your space to shine.
Features:
- Comprehensive site profiles
- Advanced search and filtering
- Interactive asset map by province
- Community discussion boards


Who Uses CCTAM?
CCTAM serves both sponsors and clinical research professionals:
For Sponsors:
- Find qualified sites fast – search by disease area, infrastructure, and location
- Reduce feasibility workload – no more chasing regional contacts
- Discover partnerships – connect with Canada’s top research talent
- Save time and money – all at no cost
For Investigators & Sites:
- Be seen by global sponsors seeking Canadian sites
- Build your network – connect with others in your field
- Unlock opportunity – grow your research pipeline
- Stay current – maintain visibility with automated record updates
A Bit of Background
CCTAM was created to streamline how trials are planned and conducted in Canada. Before its launch in 2015, site information was fragmented and often out of date. Now, CCTAM brings thousands of research assets into one place, a trusted, up-to-date national resource.
In 2023, Canada ranked 3rd globally for new clinical trials and 4th for ongoing trials.
Let’s keep that momentum going — together.
Powered by a National Network
CCTAM is hosted by N2 Canada, in partnership with Clinical Trials British Columbia, and supported by national and regional leaders in clinical research.
Evdokia Anagnostou
Organization: Holland Bloorview Kids Rehabilitation Hospital
Notes: HealthCareCAN VPR Committee
Rebecca Barnes
Organization: N2 Canada
Notes: Co-Chair
Shurjeel Choudhri
Organization: Bayer
Liz Lylyk
Organization: Research Manitoba
Ashley Hilchie
Organization: Nova Scotia Health
Stéphanie Lamarche
Organization: CATALIS Quebec
Alex Lim
Organization: ACT
Tammy Mah-Fraser
Alison Orth
Organization: Clinical Trials British Columbia
Notes: Co-Chair
Ranuka Srinivasan
Organization: Clinical Trials Ontario
Natalie Zakhem
Organization: Trade Commissioner Service
